Trends
- Feb 19, 2016
- 3 min read
Atomic radius
The atomic radius describes the size of the atom.
The atomic radius decrease when moving across a period from left to right. As we move across a period electrons are added to the same energy level and protons are added to the nucleus; increasing the effective nuclear charge and pulling the electrons closer to the nucleus.
The Atomic radius tend to increase when moving down a group from top to bottom. As we go down a group additional energy levels are added; and each energy level is further from the nucleus.

Electronegativity
Electronegativity is the measure of the tendency of an atom to attract a bonding pair of electrons. It cannot be measured directly and needs to be computed from other atomic properties.
The electronegativity increases as you move across the table from left to right and decreases as you move down the table.

Ionization energy
The ionization energy is the energy required to remove an election from a gaseous atom or ion.
In general, the 1st ionzation energy increases as we go across a period; as the electrons are held closer to the nucleus with the increasing effective nuclear charge.
In the 1st ionization energy decreases as we go down a group; as the electrons are further from the nucleus with each increasing energy level.
The noble gases possess very high ionization energies because their full valence shell makes them highly stable.

Chemical Reactivity In metals reactivity it increases as you go down a group because the farther down a group of metals you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity. In metals reactivity it decreases as you go across a period because though they still want to give away valence electrons they have more of them to get rid of, which requires more energy.

Melting Point The melting points is the amount of energy to change the solid phase of a substance to a liquid. The stronger the bond between the atoms of an element, the more energy required to break that bond. Melting points are varied and do not generally form a distinguishable trend across the periodic table. • Metals generally possess a high melting point. • Most non-metals possess low melting points. • The non-metal carbon possesses the highest boiling point of all the elements. The semi-metal boron also possesses a high melting point.
Metallic Character Metallic character decreases as you move across the periodic table from left to right. This occurs as atoms more readily accept electrons to fill a valence shell than lose them to remove the unfilled shell. Metallic character increases as you move down the periodic table. This is due to the fact that the electrons become easier to lose as the atomic radius increases.

Non-Metallic Character Non-metallic property increases across a period and decreases down the group due to the same reason as metallic properties.

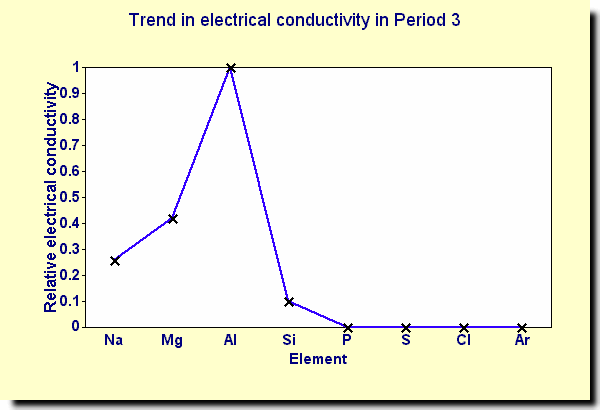
Conductivity Electrical conductivity increases going across Period 3 from sodium to aluminium, then decreases to silicon. For an element to conduct electricity, it must contain electrons that are free to move. In general, metals are good conductors of electricity and non-metals are poor conductors of electricity.
Ion Charge To find the ionic charge of an element you'll need to consult your Periodic Table. On the Periodic Table metals (found on the left of the table) will be positive. Non-metals (found on the right) will be negative.



Comments